“Bacteriophage-pesticide” golden partnership: Novel and highly effective bacteriophage LPRS20 synergizes with thiazole copper/berberine to prevent and control bacterial wilt

Literature Alert
The “Golden Combination” of Phage and Pesticide:Novel High-Efficiency Phage LPRS20 Synergistically Controls Bacterial Wilt with Thiodiazole-Copper / Berberine

【Abstract】
Phage therapy has the potential to alleviate bacterial wilt in plants. However, the lack of knowledge regarding the interaction between phages and agrochemicals has hindered the broader application of phages in agriculture. In this study, we characterized a phage isolate and investigated its interactions with agrochemicals. Anovel phage, LPRS20, belonging to the genus Ampunavirus, was identified, which exhibits a broad lytic range and significant antibacterial activity against Ralstonia solanacearum strains infecting tobacco, chili, or tomato. Sensory evaluation of tobacco leaf morphology indicated that phage application caused negligible harm to plants. Investigations into phage-agrochemical interactions revealed synergistic effects when LPRS20 was administered 4 hours prior to thiodiazole-copper, as well as when LPRS20 was combined with low-concentration berberine. Overall, our findings demonstrate that phage LPRS20 represents a novel, effective, and environmentally friendly biocontrol agent for tobacco bacterial wilt both in vivo and in vitro, contributing to the potential integration of phages and agrochemicals for the control of soil-borne pathogens.
【Research Background】
The soil-borne pathogenic bacterium Ralstonia solanacearum poses a severe threat to global agricultural production and food security, attributed to its strong virulence and extensive host range, covering more than 200 plant species across 50 plant families. Bacterial wilt of plants caused by R. solanacearum has been shown to result in extremely high yield losses in solanaceous crops. In fact, bacterial wilt is reported to cause 10-30% global yield loss in tobacco, 35-90% in tomato, and up to 100% in chili. Currently, the use of agrochemicals such as copper formulations and antibiotics remains the primary method for controlling plant bacterial wilt. However, long-term overuse of these agrochemicals has led to the widespread occurrence of agrochemical residues, bacterial resistance, and phytotoxicity. Notably, non-targeted agrochemicals may exert potential harmful effects on beneficial probiotics during plant growth and product processing, in addition to killing plant pathogens. Due to the inherent severe environmental risks associated with chemical control methods, screening novel, highly efficient, and targeted antibacterial agents to suppress plant bacterial wilt is a challenging task.
In recent years, several strategies have been developed to control plant bacterial wilt, including phage therapy, breeding disease-resistant varieties, antagonistic microorganisms, and natural antibacterial products derived from microorganisms or plants. However, the efficacy of antagonistic microbial inoculants is limited due to difficulties in rhizosphere colonization. The breeding of disease-resistant varieties relies on the exploration of disease resistance mechanisms in plants. Additionally, natural antibacterial products face stability issues, and their extraction and purification processes are complex and costly. As viruses that specifically target host bacteria, phages are abundant and easy to isolate, showing great potential in controlling plant bacterial wilt. Compared with agrochemicals, phage therapy offers advantages such as high host specificity, environmental friendliness, and self-regulated dosage in the presence of pathogenic bacteria. Previous studies have found that phage application helps inhibit pathogenic bacterial infection via lysis, enrich antibiotic-producing bacteria, and achieve a fitness trade-off between phage resistance and virulence in pathogens. Therefore, phage therapy has become a highly favored preventive and control technique for treating plant bacterial wilt.
Ralstonia solanacearum exhibits high genetic diversity and is regarded as a species complex, highlighting the necessity for continuous screening of phages targeting the Ralstonia solanacearum species complex (RSSC) to control plant bacterial wilt. Current research has reported several phages against R. solanacearum and their biocontrol potential, most of which belong to the genera Gyeongsanvirus and Serkorvirus, as well as other unclassified phages. For example, phages from the genus Gyeongsanvirus (e.g., vRsoP-WF2, vRsoP-WM2, vRsoP-WR2), phages from the genus Serkorvirus (e.g., RpTl, RpY2), and additional unclassified phages (e.g., NJ-P3, NB-P21, NC-P34, NN-P42) have all been shown to significantly alleviate symptoms of bacterial wilt in tomato. Despite these advances, existing research on R. solanacearum phages has primarily focused on tomato crops, with significant gaps in the collection and utilization of resources for other crops. This situation is unfavorable for the widespread application of phages and hinders progress in related mechanistic studies.
Recently, research on the ecological and evolutionary mechanisms of phage-mediated control of plant bacterial wilt has attracted attention, driving advances in phage application. However, empirical data verifying the safety of phages for crops remains scarce, which is crucial for developing phage-based biocontrol strategies and implementing environmentally friendly disease management strategies. Filling this knowledge gap will help promote the safe and effective application of phages in agricultural environments.
In terms of phage application strategies, monotherapy with phages may be limited due to the development of resistant bacteria, while phage-antibiotic combinations have been proven more effective than phage monotherapy in both in vitro and in vivo experiments against clinical isolates. This strategy has sparked great interest in combining phages with agrochemicals to combat soil-borne pathogens such as R. solanacearum. Phage-pesticide combinations represent a promising approach to enhance bactericidal efficacy and reduce pesticide usage in the treatment of plant pathogens, thereby promoting plant health and green agricultural development. Previous studies on phage-antibiotic interactions have facilitated the design of combination therapeutic regimens; however, to date, there is no information available on the interaction between phages and pesticides in agriculture.
This study focused on exploring novel Ralstonia phage resources from field soils planted with tobacco crops and screening the optimal phage. Subsequently, its safety and efficacy were further investigated in tobacco (Nicotiana benthamiana), a model plant for evaluating the biocontrol effect of phages on plant bacterial wilt. To further gain in-depth insights into the interaction between phages and agrochemicals, based on the isolated phage, plant-derived berberine and the metal compound thiodiazole-copper were used as representatives of agrochemicals for preliminary exploration in soil.
Results
- Phage LPRS20 exhibits a broad host range and strong inhibitory activity.
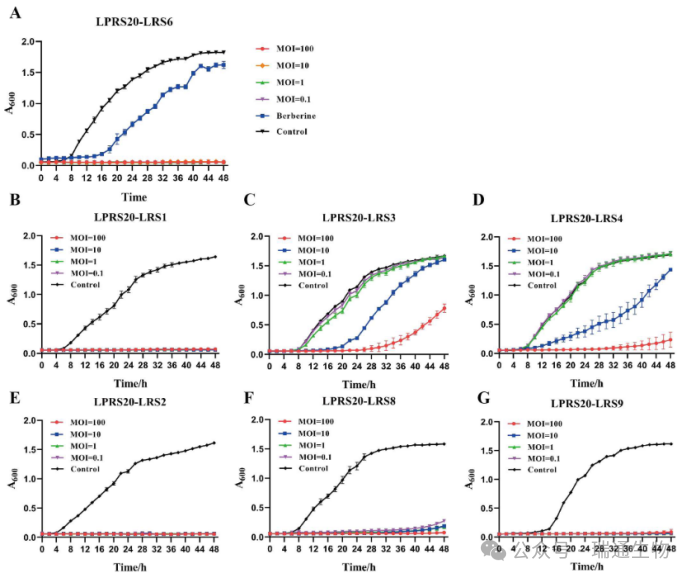
Figure 1. Inhibition curves of the isolated phage LPRS20 against R. solanacearum.In NB medium, phage LPRS20 was applied at a multiplicity of infection (MOI) ranging from 0.1 to 100 to inhibit (A) host strain LRS6 and (B) LRS1 from pepper, (C) LRS3 and (D) LRS4 from tobacco, and (E) LRS2, (F) LRS8, and (G) LRS9 from tomato.A negative control group was set up by diluting the phage solution with NB medium only.MOI: multiplicity of infection.Vertical bars represent standard deviations.
A total of 21 phages were isolated from soil samples of tobacco fields affected by bacterial wilt.Phages LPRS20 and LPRS31 formed clear plaques on plates of Ralstonia solanacearum strains from tobacco, including isolates LRS3, LRS4, and LRS6 obtained from different regions.Subsequently, the ability of LPRS20 and LPRS31 to lyse R. solanacearum isolates from other plant hosts was tested using additional strains from pepper and tomato, and the results showed that LPRS20 exhibited a broader lytic range.Furthermore, no plaques of LPRS20 were observed on beneficial microbes covering 22 species across 10 genera.Therefore, among the 21 isolated phages, LPRS20 displayed the broadest lytic spectrum against R. solanacearum and did not lyse beneficial microorganisms.
Based on its broad lytic range, phage LPRS20 was selected to evaluate its antibacterial activity.After 48 hours of incubation, the absorbance of the LPRS20-treated group (A₆₀₀ = 0.05 ± 0.01) was significantly lower than that of the 16 μg/mL berberine group (A₆₀₀ = 1.62 ± 0.06) (Figure 1A).Inhibition assays revealed that LPRS20 significantly inhibited the growth of LRS1, LRS2, LRS3, LRS4, LRS6, LRS8, and LRS9 (p < 0.05) and completely suppressed LRS1, LRS2, LRS6, and LRS9 by the end of the 48-hour detection period (Figure 1A–G).These results indicate that LPRS20 possesses strong antibacterial activity against R. solanacearum strains isolated from different host plants, including tobacco, pepper, and tomato.
- Phage LPRS20 exhibits high reproductive activity, large burst size, and strong tolerance to pH, ultraviolet radiation, and temperature
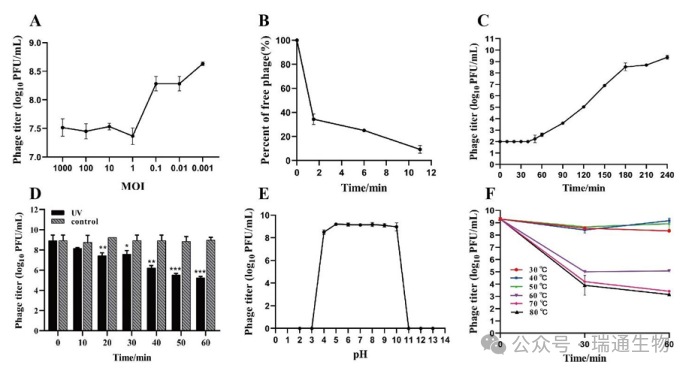
Figure S2. Biological characteristics of the isolated phage LPRS20.(A) Optimal multiplicity of infection (MOI) of LPRS20.(B) Adsorption assay of LPRS20.(C) One-step growth curve of LPRS20.(D) Ultraviolet (256 nm) stability.(E) pH stability.(F) Thermal stability.*p < 0.05; **p < 0.01; ***p < 0.001.MOI: multiplicity of infection.Vertical bars represent standard deviations.
The optimal multiplicity of infection (MOI) was determined to be 0.001, with an adsorption time of 11 minutes, laying the foundation for the one-step growth curve assay (Figure S2A, B).The one-step growth curve revealed that the average burst size of LPRS20 was 2,450 PFU per cell (Figure S2C).LPRS20 showed no significant titer loss over a wide pH range from 4.0 to 10.0.Its titer remained relatively stable after incubation at temperatures up to 50 °C for 60 minutes.In addition, although the phage titer decreased significantly, LPRS20 remained infectious after exposure to ultraviolet (UV₂₅₄ nm) radiation for 60 minutes (Figure S2D–F).These results indicate that LPRS20 possesses high reproductive activity and a large burst size, while exhibiting remarkable stability across a wide range of pH, UV, and temperature conditions.
- The lytic phage LPRS20 represents a novel species belonging to the genus Ampunavirus
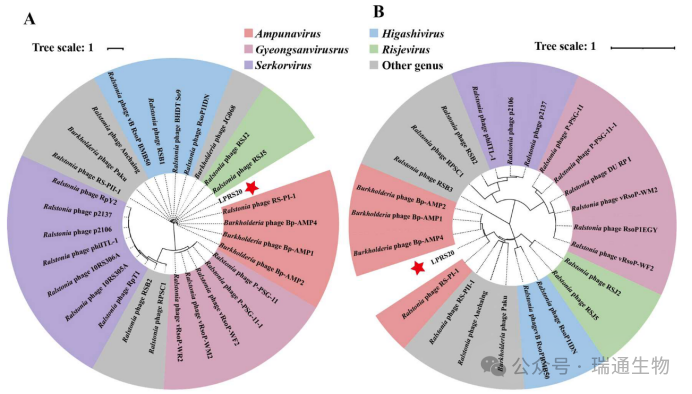
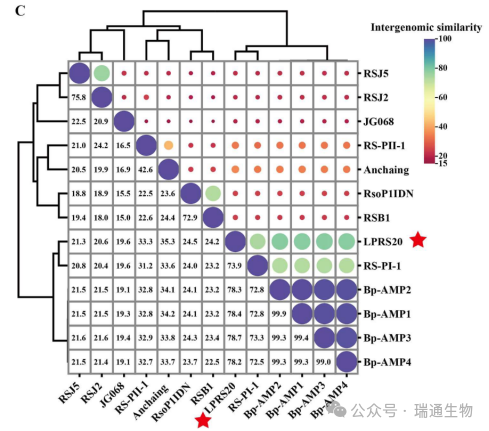
Figure 2. Phylogenetic analysis of the closely related phages and phage LPRS20 based on the protein sequences of (A) major capsid protein and (B) large terminase subunit.Members of the genera Ampunavirus, Gyeongsanvirus, Serkorvirus, Risjevinus, and Higashivinus are distinguished by different colors.(C) Intergenomic similarity comparison between phage LPRS20 and other phages in Figure S4.The red star indicates the isolated phage LPRS20.
Whole-genome analysis revealed that the genome of phage LPRS20 consists of double-stranded DNA with a length of 43,060 bp and a G+C content of 62%.A total of 50 open reading frames (ORFs) were predicted, and putative functions were annotated for 26 of these ORFs.These ORFs were classified into four functional modules, including phage structural proteins, nucleotide metabolism and genome replication, cell lysis, and unknown functions (Figure S3).In addition, no homologs of phage virulence factors (VFs) were predicted, and no homologs of antibiotic resistance genes (ARGs) were present in the genome.PhaTYP analysis indicated that LPRS20 is a lytic phage.
Based on whole-genome alignment results (Table S3), we infer that LPRS20 belongs to the genus Ampunavirus.Ampunavirus phages, including Ralstonia phage RS-PI-1 and Burkholderia phage Bp-AMP1, showed high similarity to LPRS20, with shared protein identities of 82.00% and 68.00%, respectively (Table S4).This result further supports the hypothesis that LPRS20 may belong to the genus Ampunavirus.
Phylogenetic trees of representative Ralstonia and Burkholderia phages were constructed based on the amino acid sequences of the major capsid protein and large terminase subunit.A distinct cluster contained only Ampunavirus phages, including LPRS20, Burkholderia phages Bp-AMP1, Bp-AMP2, and Bp-AMP4, and Ralstonia phage RS-PI-1 (Figure 2A, B).The intergenomic similarity between LPRS20 and Burkholderia phage Bp-AMP4 of the genus Ampunavirus reached 78.7% (Figure 2C).Therefore, LPRS20 is classified as a novel species within the genus Ampunavirus.
4.In Vitro and In Vivo Biocontrol Activity of Phage LPRS20 Against Ralstonia solanacearum
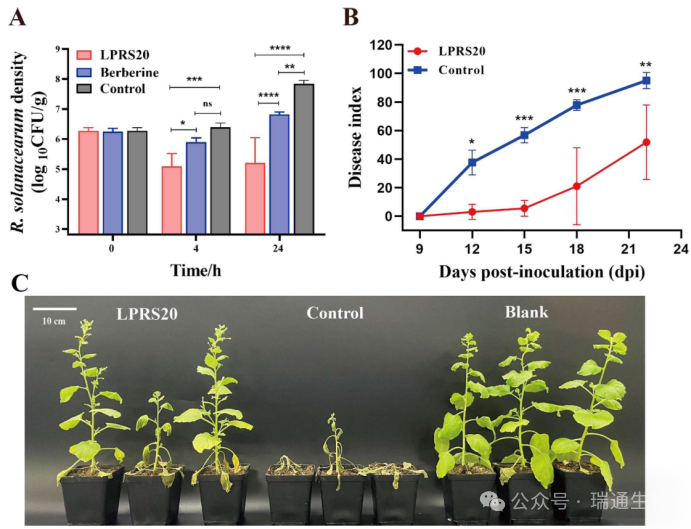
Figure 3. Biocontrol assays of phage LPRS20 in soil and tobacco plants.(A) Biocontrol activity of LPRS20 against R. solanacearum strain LRS6 in soil.(B) Biocontrol efficacy of LPRS20 against tobacco bacterial wilt.(C) Observation of tobacco plants at 22 days post-inoculation.The blank control group was treated with PBS.Significance is indicated as *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001; ns, not significant.Vertical bars represent standard deviations.
To evaluate the biocontrol activity of LPRS20 in soil, a bactericidal assay was performed against R. solanacearum in soil.After 4 hours of incubation, application of phage LPRS20 significantly reduced (p < 0.05) the viable count of R. solanacearum strain LRS6 by 1.30 ± 0.51 log₁₀ CFU/g compared with the control group, and by 0.81 ± 0.32 log₁₀ CFU/g compared with the berberine-treated group.After 24 hours of incubation, treatment with LPRS20 significantly decreased the viable count of R. solanacearum by 2.64 ± 0.74 log₁₀ CFU/g (p < 0.0001) compared with the control group, and by 1.62 ± 0.82 log₁₀ CFU/g compared with the berberine-treated group (Figure 3A).
To further assess the efficacy of LPRS20 in controlling R. solanacearum infection in vivo, a tobacco bacterial wilt model was established.Compared with the control group, application of LPRS20 significantly alleviated bacterial wilt symptoms in tobacco (Figure 3C).The disease index (DI) of tobacco showed that the DI values of the LPRS20 group were significantly lower than those of the control group at 12, 15, 18, and 22 days post-inoculation (p < 0.05).At 22 days post-inoculation, the DI of the LPRS20 group decreased from 95.06 to 51.85 compared with the control group, showing an obvious biocontrol efficacy of 45.46% (Figure 3B).Therefore, phage LPRS20 can effectively control tobacco bacterial wilt.
5.In Vitro Combined Effects of Phage and Pesticides
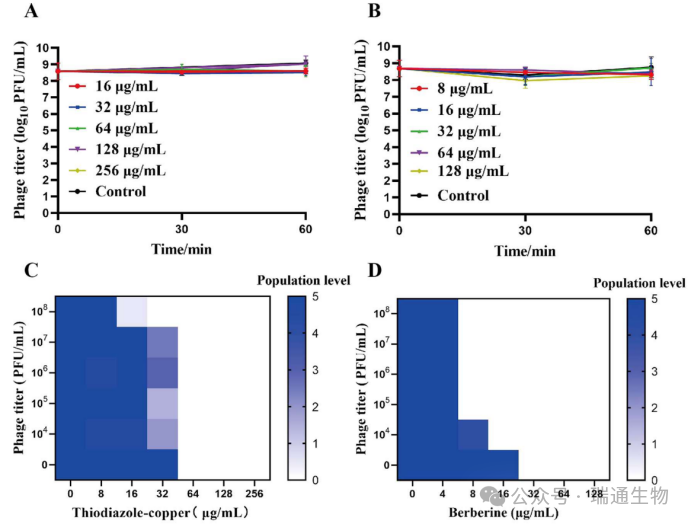
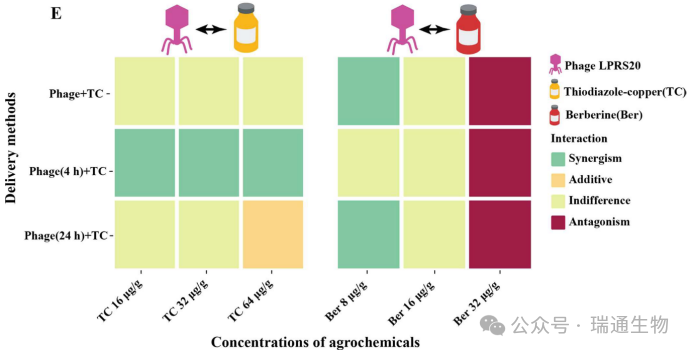
Figure 4. Combined effects of the isolated phage LPRS20 with thiodiazole‑copper or berberine.Stability of LPRS20 in (A) thiodiazole‑copper and (B) berberine.Inhibition of LRS6 growth by combinations of LPRS20 with (C) thiodiazole‑copper or (D) berberine in NB medium in 96‑well plates.(E) Interactions between phage LPRS20 and thiodiazole‑copper or berberine.For more details on the criteria for judging interactions, see Tables S10 and S11.
Tolerance of phage LPRS20 to pesticides contributes to a preliminary understanding of the effects of pesticides on phages.Therefore, phage titers were determined after incubation with six concentrations of thiodiazole‑copper or berberine for 60 minutes.The results showed that phage LPRS20 was relatively stable, and no significant titer loss was observed at any concentration of thiodiazole‑copper or berberine (Figure 4A, B).This indicates that the activity of LPRS20 is not affected by thiodiazole‑copper and berberine.
To further investigate the combined effects of phages and pesticides against R. solanacearum, the population levels of R. solanacearum treated with combinations of phage plus thiodiazole‑copper or berberine were determined in vitro.In cells treated with LPRS20 alone, a high population level of grade 5 (10⁴ to 10⁶ PFU/mL) was observed.However, the combination of 10⁸ PFU/mL LPRS20 with >16 μg/mL thiodiazole‑copper almost completely reduced the number of viable cells (population level ≤ 1) (Figure 4C).Under LPRS20 combined with berberine treatment, bacterial cells were completely inhibited (population level = 0) when R. solanacearum was exposed to >8 μg/mL berberine plus LPRS20 in the range of 10⁵ to 10⁸ PFU/mL (Figure 4D).
Overall, through various combinations of different concentrations of phages and pesticides, the lowest possible concentrations of thiodiazole‑copper (16 μg/mL) and berberine (8 μg/mL) could be identified to achieve nearly complete killing efficacy.
6.The Type, Application Mode, and Concentration of Pesticides Determine Their Interaction Patterns with PhagesBased on the nearly complete killing effect observed in the heatmap, we selected three different concentration combinations of LPRS20 + agrochemicals to determine their killing efficacy against R. solanacearum in soil, thereby clarifying the interactions between phages and agrochemicals (Figure 4C, D).
For the combination of LPRS20 and thiodiazole‑copper, the application mode of phage (4 h) + thiodiazole‑copper produced a synergistic effect, with a significant reduction in bacterial counts compared with the phage‑only and thiodiazole‑copper‑only groups (p < 0.05) (Figure 4E and S5A).However, the delivery modes of phage + thiodiazole‑copper and phage (24 h) + thiodiazole‑copper showed indifferent or additive effects in the combined treatments.
For the LPRS20 + berberine combination, the combination of LPRS20 + 8 μg/g berberine produced synergistic effects under both phage + berberine and phage (24 h) + berberine application modes.The number of viable bacteria in the combination groups was significantly reduced compared with the phage‑only and berberine‑only groups (p < 0.05) (Figure 4E and S5B).When the berberine concentration was increased to 16 μg/g, the phage‑berberine interaction was indifferent.Subsequently, antagonism was observed in the combination of LPRS20 with a high dose of 32 μg/g berberine.
Overall, the application mode of phage (4 h) + thiodiazole‑copper resulted in synergism in LPRS20 + thiodiazole‑copper treatments, while the combination of LPRS20 with a low concentration of 8 μg/g berberine also produced synergism.The results also indicate that the interaction between phage and thiodiazole‑copper differs from that between phage and berberine.
7.Phage LPRS20 Is Non‑Phytotoxic to Tobacco

Figure 5. Tobacco leaves collected from the same position of plants after 72 hours of treatment with three concentrations of phage LPRS20, thiodiazole-copper, and berberine, respectively.
Phages are regarded as safe agents for pathogen control, whereas traditional agrochemicals may be phytotoxic to plants.Therefore, we evaluated phytotoxic symptoms by treating tobacco leaves sequentially with LPRS20, thiodiazole‑copper, and berberine via sensory assessment, to explore differences in the effects of phages and agrochemicals on plants.
Phytotoxic symptoms including spotting, yellowing, and curling appeared on tobacco leaves treated with thiodiazole‑copper at 64 or 128 μg/mL, while wilting was observed in leaves treated at 128 μg/mL (Figure 5).Compared with the control group, treatments with 64 and 128 μg/mL thiodiazole‑copper resulted in significant spotting, yellowing, and curling.Similarly, wilting symptoms were also significant after treatment with 128 μg/mL berberine compared with PBS treatment.In contrast, no significant differences in symptoms were observed between the LPRS20‑treated and control groups (Table 2).
Overall, symptoms on tobacco leaves after phage treatment indicated that phages caused no damage to tobacco, in sharp contrast to the effects of thiodiazole‑copper and berberine.
Discussion
In this study, a novel and efficient phage, LPRS20, was isolated and screened from 21 candidate phages.Phylogenetic analysis revealed that LPRS20 belongs to a new species within the genus Ampunavirus, designated Ampunavirus LPRS20.As a phage, LPRS20 shows great potential for the biocontrol of various plant bacterial wilt diseases due to its broad lytic range, remarkable antibacterial activity, strong reproductive activity (with a large burst size), and significant biocontrol efficacy.
Phage LPRS20 exhibits tolerance to a wide range of pH, ultraviolet radiation, temperature, and pesticides, indicating excellent environmental compatibility.Meanwhile, LPRS20 does not harm tobacco leaves, lacks virulence factors (VFs) and antibiotic resistance genes (ARGs), and shows no infectivity toward beneficial microorganisms in the plant rhizosphere, highlighting its value as an environmentally friendly biocontrol agent.
The study also confirmed that, compared with pesticides, phages cause negligible harm to plants and display high target specificity.Furthermore, exploration of phage application strategies revealed that adding thiodiazole‑copper 4 hours after phage administration produced synergistic effects in phage + thiodiazole‑copper combinations, while better efficacy was achieved when phages were combined with a low concentration of 8 μg/g berberine.
Several hypothetical synergistic mechanisms may underlie the synergistic effects between phages and pesticides:
1.Multi-target Attack HypothesisPhages specifically recognize receptors on the bacterial surface, inject genetic material, replicate using host machinery, and eventually lyse bacterial cells.Thiodiazole‑copper / berberine: interfere with bacterial metabolism, disrupt cell membrane integrity, and inhibit the activity of key enzymes.Synergistic effect: the two agents act on distinct targets, making it difficult for bacteria to develop simultaneous multiple resistances.
2.Sublethal Injury Sensitization HypothesisPesticides at subinhibitory concentrations cause damage to bacterial cell membranes and metabolic stress.Bacteria injured in this way become more susceptible to phage adsorption and infection.Accelerated phage replication further enhances the bactericidal effect of pesticides.
3.Biofilm Disruption HypothesisRalstonia solanacearum can form biofilms to protect itself.Pesticides may disrupt biofilm structure and expose embedded bacteria, allowing phages to more easily access and infect the exposed bacterial cells.
Implications
This study successfully isolated and identified a novel, efficient and broad‑spectrum Ralstonia solanacearum phage LPRS20, and systematically revealed its synergistic mechanisms with thiodiazole‑copper and berberine for the first time.The results demonstrate that rational combined application of phages and pesticides can achieve the control goal of “reducing pesticides without compromising efficacy, enhancing activity while being more eco‑friendly”, providing a new strategy for the green control of bacterial diseases such as bacterial wilt.Through scientific compatibility to exert their respective advantages, a multi‑target and multi‑level integrated control system can be established, which represents the inevitable path toward sustainable agricultural development.
Translated from:Exploration of Phage‑Agrochemical Interaction Based on a Novel Potent Phage LPRS20‑Targeting Ralstonia solanacearum
Compiled by: Gao Zhixiao