Engineered phage with cell-penetrating peptides for intracellular bacterial infections

Abstract
A research team from the Children’s Hospital of Chongqing Medical University, the Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences, and other institutions has published a paper in mSystems entitled Engineered Phage with Cell-Penetrating Peptides for Combating Intracellular Bacterial Infections. Focusing on infections caused by Salmonella, a typical intracellular pathogen, and the growing clinical challenge of antibiotic resistance, this study developed an engineering strategy based on non-model bacteriophages. Using CRISPR-Cas9 technology, cell-penetrating peptides were displayed on the surface of GP94, an Ig-like domain protein of the Salmonella phage selz, which significantly improved phage internalization efficiency and enabled highly effective inhibition of intracellular Salmonella. This work established a universal and convenient engineering method for non-model phages, providing a feasible and highly efficient novel technical route for phage therapy against intracellular drug-resistant bacterial infections.
Introduction
As a typical facultative intracellular pathogen, Salmonella can invade a variety of host cells and proliferate intracellularly. Its intracellular localization allows it to evade both antibiotics and host immune surveillance. Coupled with the continuous emergence and spread of drug-resistant strains, the clinical treatment of Salmonella infections has become considerably more difficult, posing a major threat to global public health.
Phage therapy has emerged as a promising alternative to combat drug-resistant bacteria due to its high host specificity, minimal disruption to the normal microbiota, in situ replication at infection sites, and low toxicity. However, the low cellular uptake efficiency of natural phages severely limits their ability to target intracellular pathogens, restricting their application in the treatment of intracellular bacterial infections.
Although cell-penetrating peptides (CPPs) can markedly enhance the intracellular delivery efficiency of macromolecules and have shown potential for improving phage internalization in model phage engineering, progress in the engineering of non-model phages has been slow. This is largely due to limited understanding of non-model phage structures and the lack of robust strategies for surface functional peptide display.
Meanwhile, immunoglobulin-like (Ig-like) domains on phage surfaces are often exposed externally, representing potential sites for peptide display, yet they have not been systematically applied to the engineering of non-model Salmonella phages.
Against this background, the present study aimed to:
Screen and verify the feasibility of GP94, an Ig-like domain protein of the Salmonella phage selz, as a surface display site for exogenous peptides;
Establish a CRISPR-Cas9-based genome editing technique for non-model phages to achieve efficient display of CPPs on GP94;
Construct engineered phages with significantly improved intracellular uptake efficiency and screen for the optimal CPP modification scheme;
Evaluate their inhibitory effect on intracellular Salmonella and biosafety in mammalian cells.
Ultimately, this study established a universal and convenient engineering strategy for non-model phages, providing a novel and highly efficient therapeutic approach for intracellular and drug-resistant bacterial infections.
Results
(I) Screening and Validation of Phage Protein Candidates for Peptide Display
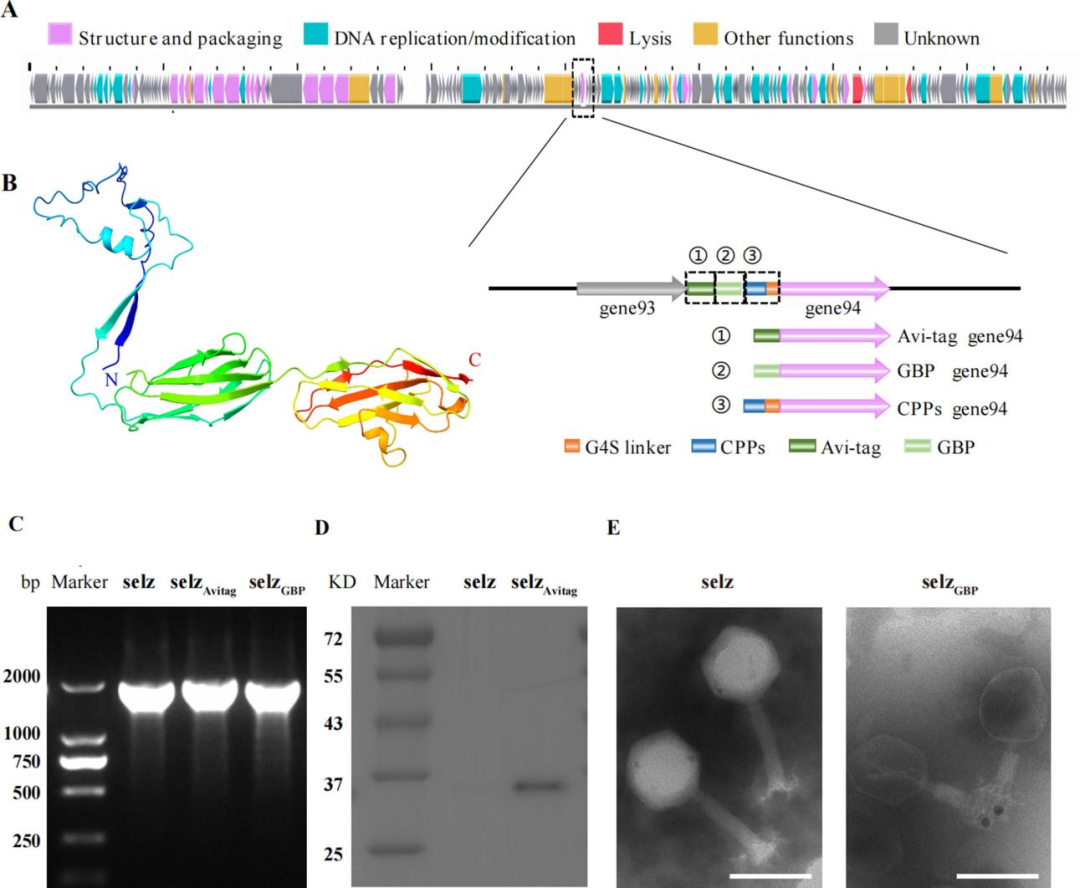
Through bioinformatic analysis, the GP94 protein containing an Ig-like domain was identified from the Salmonella phage selz. AlphaFold2 prediction showed that GP94 consists of three domains and shares high homology with the RB49 Hoc protein and the λ phage gpV protein, both of which are known to be applicable for phage display.
The Avi tag and a gold-binding peptide (GBP) were separately fused to the N-terminus of GP94 using CRISPR-Cas9 technology. Western blot analysis detected signals of biotinylated GP94 protein. Transmission electron microscopy (TEM) observed 10 nm gold nanoparticles bound to the tail of the selzGBP engineered phage, whereas no such binding was observed in the wild-type phage. These results confirm that GP94 can serve as a reliable site for peptide display.
Figure 1 Screening and validation of phage protein candidates for peptide display
(II) Biological Characteristics of Engineered Phages Fused with CPPsSeven highly efficient cell‑penetrating peptides (CPPs) for intracellular delivery were selected and fused to GP94 via CRISPR‑Cas9 to construct engineered phages. Double‑layer agar assays showed that both engineered phages and wild‑type selz formed similar translucent plaques. One‑step growth curves revealed that both had a latent period of 30 min and reached the plateau phase at 50 min, with burst sizes ranging from 57 to 104 PFU per infected cell, showing no statistically significant differences. These results confirm that CPP fusion does not affect the fundamental biological characteristics of the phage.
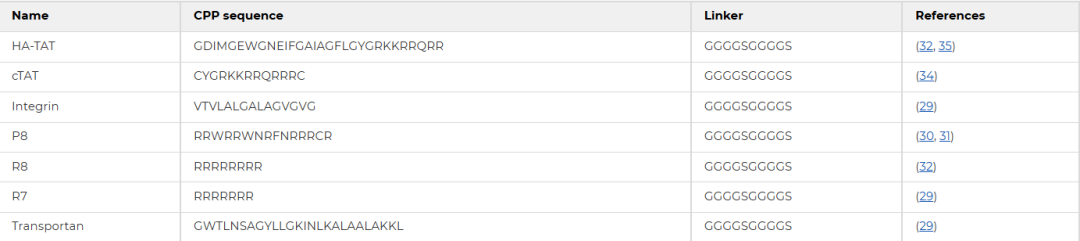
Table 1 Amino acid sequence information of CPPs
(III) CPPs Enhance the Uptake Efficiency of Phages in Mammalian CellsWild-type and engineered phages were co-incubated with HeLa, A549, Caco-2, THP-1 and Raw264.7 cells, respectively. Quantitative plaque assays showed that:In HeLa cells, the uptake of selz<sup>HA-TAT</sup> and selz<sup>Transportan</sup> was significantly increased;In A549 cells, the uptake of selz<sup>HA-TAT</sup>, selz<sup>Transportan</sup>, selz<sup>Integrin</sup> and selz<sup>R7</sup> was higher than that of the wild-type phage;In Caco-2 cells, only selz<sup>HA-TAT</sup> exhibited significantly elevated uptake, which was also the highest among all tested epithelial cells.
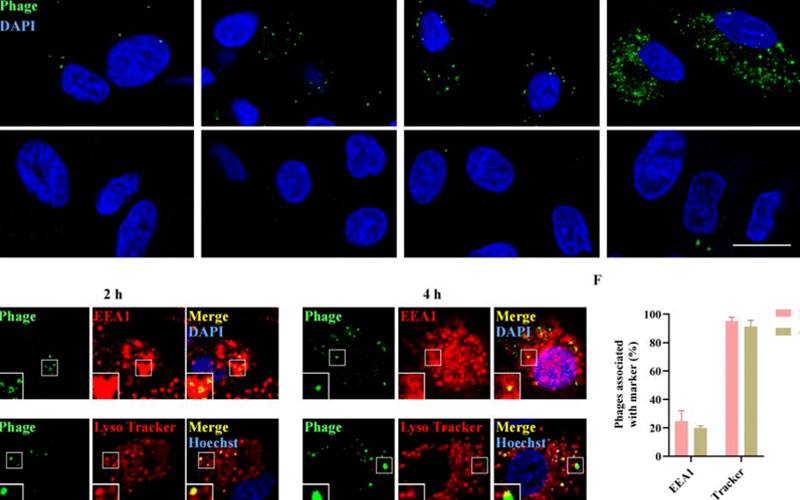
Confocal microscopy confirmed that fluorescent aggregates appeared intracellularly after 1 h of incubation with selz<sup>HA-TAT</sup> and continued to increase within 24 h, with internalization mainly mediated by the endocytosis‑lysosome pathway
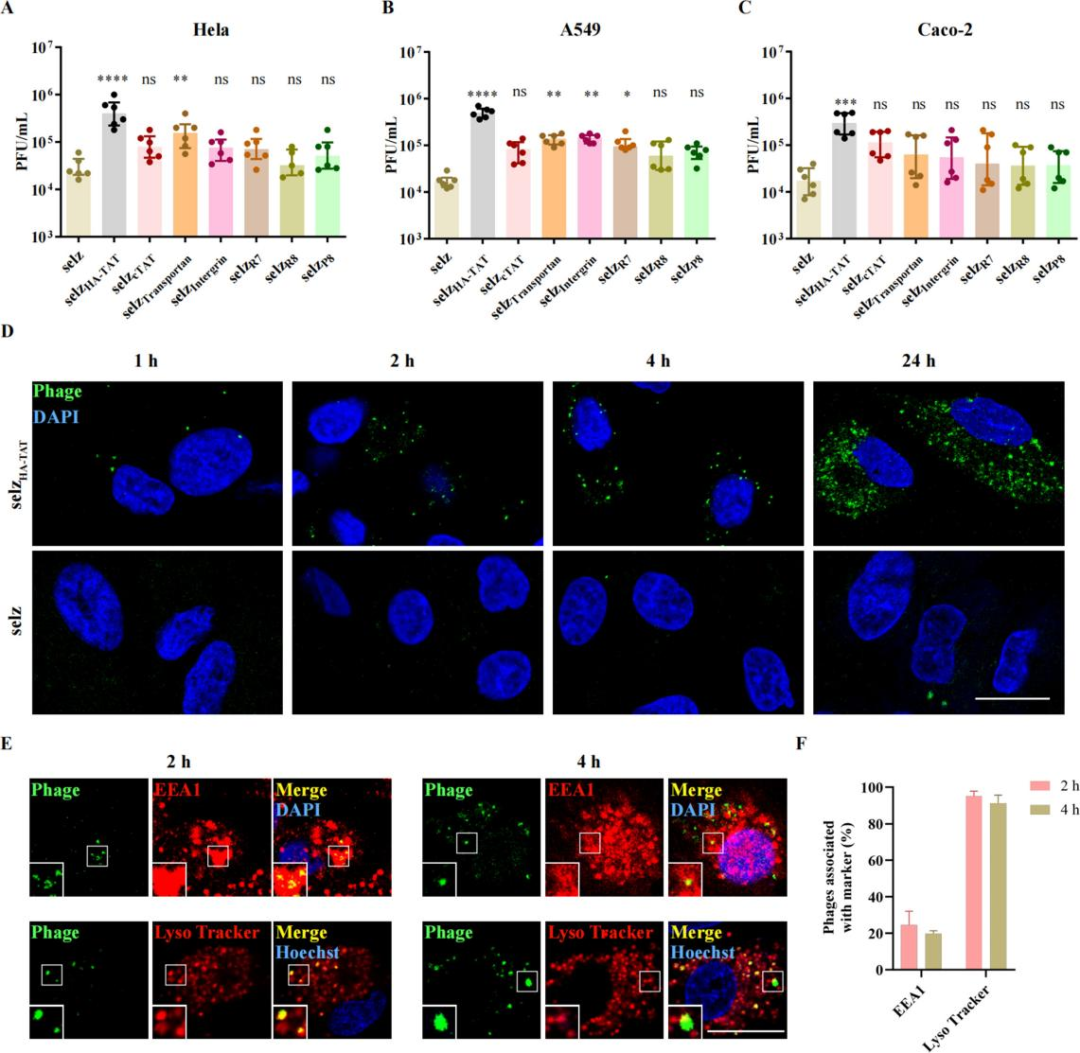
Figure 2 Cellular uptake of wild-type and engineered selz phages in different cell lines
(IV) Characterization of Engineered Phage selz<sup>HA-TAT</sup> and Its Intracellular Bactericidal EffectTransmission electron microscopy showed that selz<sup>HA-TAT</sup> exhibited identical morphology to wild-type selz (width approximately 86 nm, length approximately 95 nm, contractile tail length approximately 113 nm). In vitro lysis efficiency against Salmonella was comparable at different MOIs.
Intracellular bactericidal assays demonstrated that treatment with selz<sup>HA-TAT</sup> significantly reduced intracellular bacterial load in HeLa cells, with a bactericidal efficiency of 64%, and 48% in A549 cells, whereas no significant inhibitory effect was observed in Caco-2 cells. Flow cytometry confirmed an 18% decrease in mCherry fluorescence intensity of intracellular Salmonella in HeLa cells, providing direct evidence for effective elimination of intracellular bacteria.
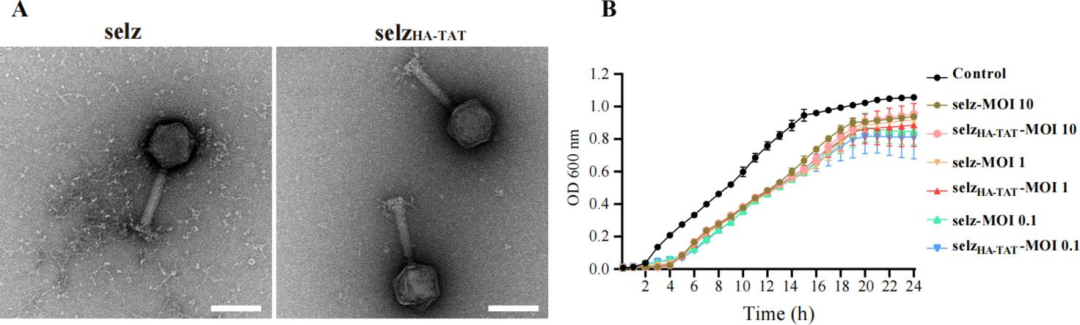
Figure 3 Characterization of wild-type selz and selz<sup>HA-TAT</sup>
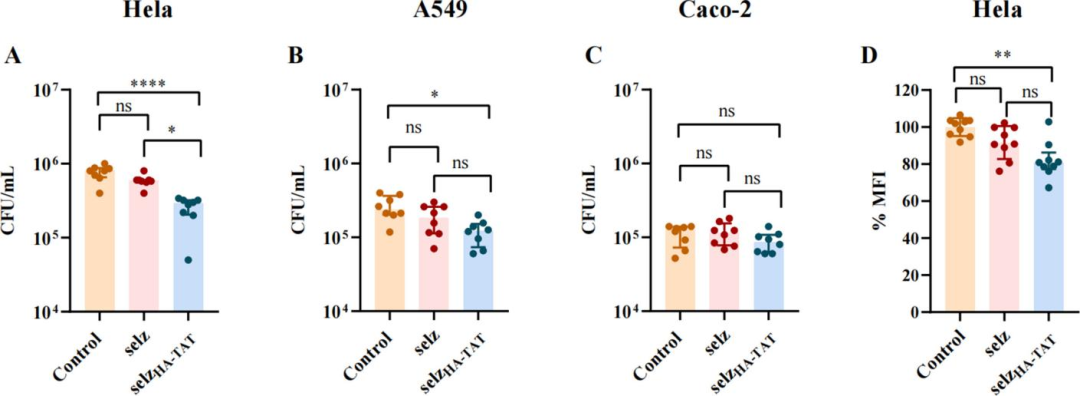 Figure 4 Bactericidal effect of selz<sup>HA-TAT</sup> on intracellular Salmonella in three epithelial cell lines
Figure 4 Bactericidal effect of selz<sup>HA-TAT</sup> on intracellular Salmonella in three epithelial cell lines
(V) Evaluation of Antibacterial Efficacy In VitroThe cytotoxicity of selz<sup>HA-TAT</sup> against HeLa, A549 and Caco-2 cells was measured using the LDH assay. The results showed no significant difference in LDH release between the selz<sup>HA-TAT</sup> treatment group, the control group and the wild-type phage group. This confirms that the engineered phage exhibits no obvious cytotoxicity to mammalian cells and possesses favorable biosafety
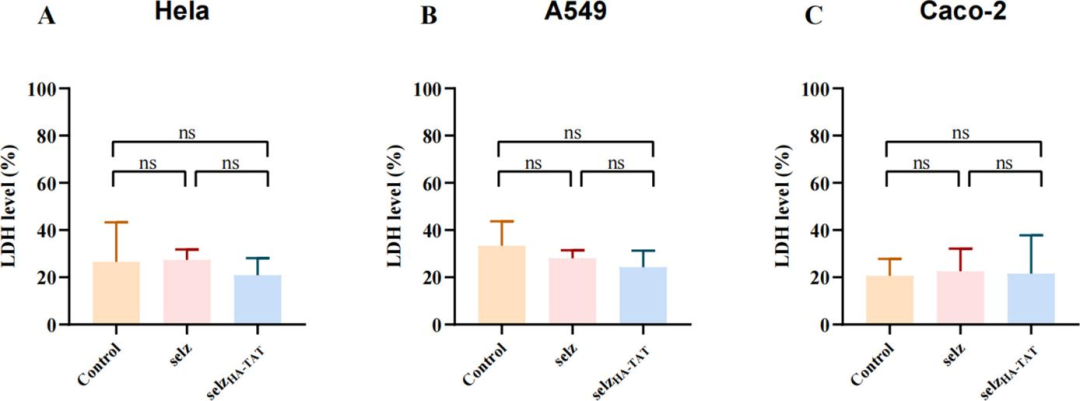 Figure 5 Cytotoxicity of wild-type phage and selz<sup>HA-TAT</sup>
Figure 5 Cytotoxicity of wild-type phage and selz<sup>HA-TAT</sup>
Conclusion
In this study, GP94 protein from Salmonella phage selz was successfully identified as a foreign peptide display site, and a CRISPR-Cas9-based engineering method for non-model phages was established. By displaying the cell-penetrating peptide HA-TAT, the engineered phage selz<sup>HA-TAT</sup> with significantly improved intracellular uptake efficiency was obtained. This phage can enter epithelial cells via the endocytic pathway, efficiently kill intracellular Salmonella, and shows no toxicity to mammalian cells.
The study confirms that Ig-like domains can serve as universal display sites on the surface of non-model phages, and CPP fusion is an effective strategy to enhance phage internalization efficiency. It provides a novel therapeutic approach for intracellular drug-resistant bacterial infections and is expected to address the global antibiotic resistance crisis.
Implications
This study offers multiple important insights for phage engineering and the treatment of intracellular bacterial infections:
From a scientific and technical perspective
Breaking through technical bottlenecks and expanding engineering scopeIt verifies the considerable engineering potential of non-model phages. Precise gene editing via CRISPR-Cas9, combined with the universal display strategy of Ig-like domains, successfully overcomes the previous limitation that only model phages could be modified.
Solving the targeting challengeFusion of cell-penetrating peptides with phages effectively resolves the low internalization rate and poor cell entry ability of natural phages, enabling precise targeting of intracellular pathogens.
From an industrial development perspective
Opening a new blue-ocean market for anti-infective therapies and filling treatment gapsThis strategy can be directly extended to the treatment of refractory intracellular pathogens such as Mycobacterium tuberculosis and Staphylococcus aureus, providing a highly promising alternative therapy for the global fight against superbugs and drug-resistant bacteria.
Empowering the health and animal health industriesIt promotes the cross-application and practical implementation of engineered phage therapy in both human medicine and veterinary medicine, expanding the commercial potential of the entire phage industry.
This article is translated from:Engineered Phage with Cell-Penetrating Peptides for Intracellular Bacterial Infections
Compiled by: Dong Yabing